The Problem with FDA’s Warning to Abiomed

This week in Pedersen’s POV, our senior editor addresses a controversial FDA warning letter and its broader implications for medical device manufacturers.
Read the full article at: www.mddionline.com
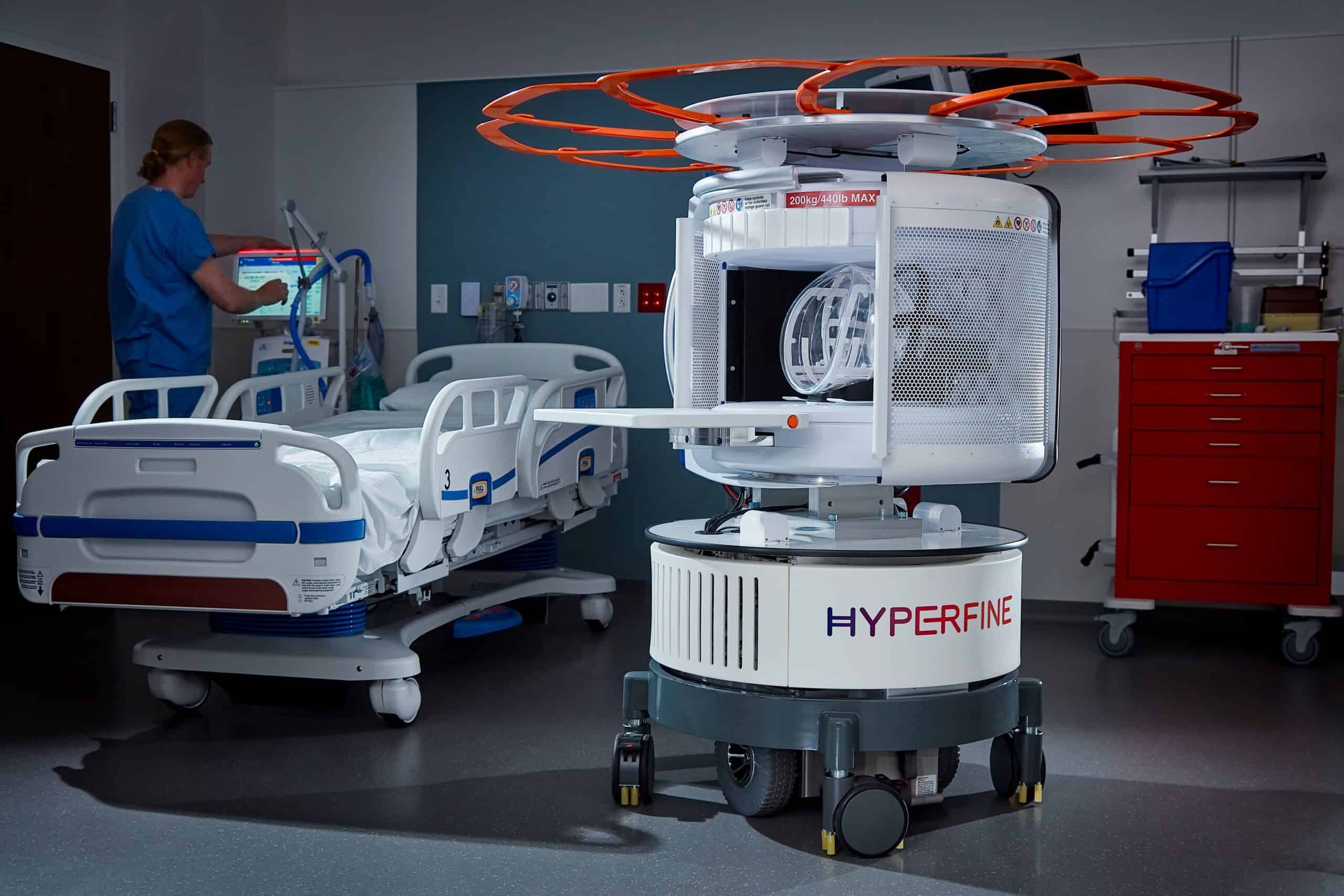
Continue Reading »Hyperfine wins FDA nod to expand MRI image-sharpening AI
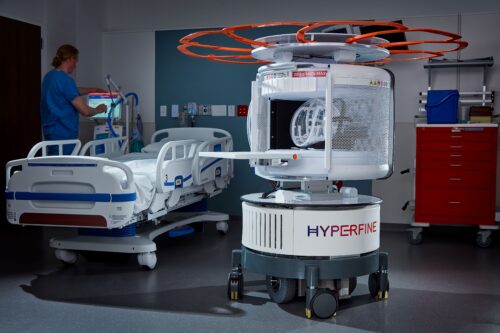
The latest nod marks Hyperfine’s eighth FDA clearance in the span of three years for its MRI scanner-on-wheels.
Read the full article at: www.fiercebiotech.com
Continue Reading »FDA to form advisory committee for digital health and AI

The FDA is enlisting a new panel of outside experts to help it get deeper into the AI weeds.
Read the full article at: www.fiercebiotech.com
Continue Reading »New tool reveals how drugs affect men, women differently — and will make for safer medications

Researchers have developed a powerful new tool to understand how medications affect men and women differently, and that will help lead to safer, more effective drugs in the future.
Read the full article at: www.sciencedaily.com
Continue Reading »MRI-Compatible Stereotactic Neurosurgery Robot

Dr. Gregory Fischer, in his keynote at BIOMEDevice Boston, discusses the journey from concept to commercialization for his MRI-compatible neurosurgery robot.
Read the full article at: www.mddionline.com
Continue Reading »The Approvals Keep Rolling in for Boston Scientific

The Marlborough, MA-based company has won approval for the latest generation of its Watchman LAAC device.
Read the full article at: www.mddionline.com
Continue Reading »Assessing the Cost & Regulatory Demands Cardiac Implantables

Mayo Clinic’s Marie Reyes discusses the…
Read the full article at: www.mddionline.com
Continue Reading »Apellis Announces U.S. FDA Approval of the EMPAVELI® Injector, a Device to Streamline Self-Administration

Apellis Announces U.S.FDA Approval of the EMPAVELI® Injector, a Device to Streamline Self-Administration – read this article along with other careers information, tips and advice on BioSpace…
Read the full article at: www.biospace.com
Continue Reading »NIH, Industry Partner on Pediatric Medical Device Advancement

They will develop a national pediatric medical device ecosystem to narrow the gap in availability, options, and innovation seen in their adult counterparts.
Read the full article at: www.mddionline.com
Continue Reading »FDA Finalize Human Factors Guidance in Combination Medical Devices

The guidance focuses on applying HFE principles for medical devices combined with a drug or biological product.
Read the full article at: www.mddionline.com
Continue Reading »