Scoop.it
Apple may nix pulse ox sensor from Apple Watch, Masimo says
Posted on Leave a Comment

The import ban affects only domestic sales of the Series 9 and Ultra 2 models of the Apple Watch, which Apple briefly stopped selling in December.
Read the full article at: www.fiercebiotech.com
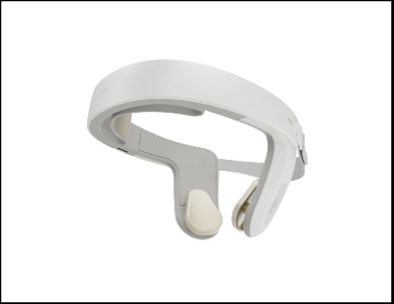
Continue Reading »Nexalin Technology Unveils Next-Generation HALO™ Clarity
Posted on Leave a Comment

Nexalin Technology Unveils Next-Generation HALO™ Clarity – read this article along with other careers information, tips and advice on BioSpace…
Read the full article at: www.biospace.com
Continue Reading »Vaporized Hydrogen Peroxide Sterilization Gets FDA Established Category A Status
Posted on Leave a Comment

An Established Category A sterilization method has a long history of safe and effective use on medical devices, putting VHP on the same level as EtO and radiation.
Read the full article at: www.mddionline.com
Continue Reading »Abbott launches first human trials of its pulsed field ablation system for afib as the sector grows
Posted on Leave a Comment
Abbott announced it has begun conducting the first human procedures using its new Volt system, through a study of more than 30 patients in Australia.
Read the full article at: www.fiercebiotech.com
Continue Reading »FDA approves Medtronic’s rechargeable deep brain stimulator for Parkinson’s, epilepsy and more
Posted on Leave a Comment

Barely a month after Medtronic announced the European clearance of its rechargeable Percept RC neurostimulator, it has racked up a U.S. approval, too.
Read the full article at: www.fiercebiotech.com
Continue Reading »Nanowear collects FDA OK for wearable AI blood pressure monitor
Posted on Leave a Comment

The maker of a wearable garment for tracking heart rate, breathing and physical activity has obtained an FDA clearance for artificial intelligence-powered software that enables continuous monitoring of blood pressure without an inflatable cuff.
Read the full article at: www.fiercebiotech.com
Continue Reading »FDA grants first clearance to AI program for diagnosing idiopathic pulmonary fibrosis
Posted on Leave a Comment
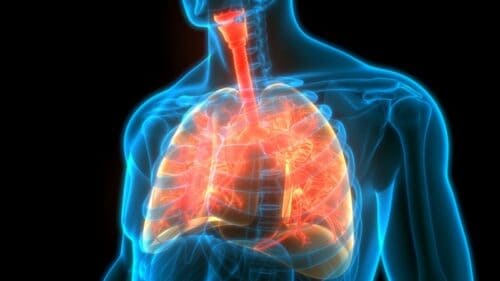
The AI developer Imvaria has claimed a de novo clearance from the FDA for a digital diagnostic that analyzes chest CT scans for the signs of IPF.
Read the full article at: www.fiercebiotech.com
Continue Reading »Congressional Watchdog to Launch Probe into FDA Medical Device Recall Oversight
Posted on Leave a Comment

Two prominent senators called for an inquiry into the agency’s handling of device recalls through an open letter published last month.
Read the full article at: www.mddionline.com
Continue Reading »Cognito opens biomarker study of neuromod device for Alzheimer’s
Posted on Leave a Comment

As it races toward FDA review of its light- and sound-based neuromodulation device, Cognito Therapeutics is taking an even closer look at the tech.
Read the full article at: www.fiercebiotech.com
Continue Reading »FDA clears vibrating belt to boost brittle bones in women facing osteoporosis
Posted on Leave a Comment

The FDA has granted a clearance to its first prescription medical device to help treat low bone density, with a wearable vibrating belt designed for postmenopausal women.
Read the full article at: www.fiercebiotech.com
Continue Reading »