Scoop.it
Omeq Medical bags FDA approval for epidural guidance device
Posted on Leave a Comment
A medical device that aims to make administration of epidurals more reliable and safer has been cleared by the FDA, giving its developer Omeq Medical its first approved product.
Read the full article at: www.fiercebiotech.com
Continue Reading »Smart Toilet Seat Wins FDA Nod to Monitor Heart Rate
Posted on Leave a Comment

Casana said it will also pursue additional filings including systolic and blood pressure.
Read the full article at: www.mddionline.com
Continue Reading »Top Mistakes in Medical Product Development
Posted on Leave a Comment

Underestimating the importance of human factors, funding, and classification can be the difference between product success and failure.
Read the full article at: www.mddionline.com
Continue Reading »Spotting Problems in 510(k) Submissions to Avoid Device Recalls
Posted on Leave a Comment

A study from the University of Minnesota offers guidance on how to avoid safety recalls for 510(k) devices.
Read the full article at: www.mddionline.com
Continue Reading »Great Britain to Delay New Medical Device Regulations
Posted on Leave a Comment

The UK government is now aiming to implement new medical device regulations for the Great Britain market in July 2025, pending Parliamentary approval.
Read the full article at: www.mddionline.com
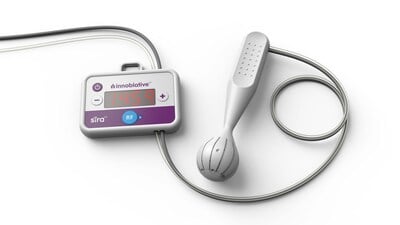
Continue Reading »Innoblative Receives U.S. FDA Breakthrough Device Designation for its SIRA RFA Electrosurgical Device
Posted on Leave a Comment
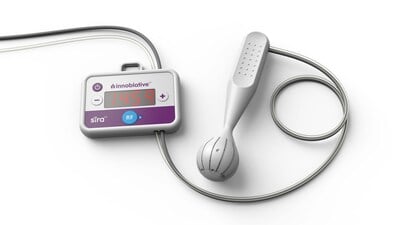
Innoblative Receives U.S.FDA Breakthrough Device Designation for its SIRA RFA Electrosurgical Device – read this article along with other careers information, tips and advice on BioSpace…
Read the full article at: www.biospace.com
Continue Reading »What is the Minimum Log-Reduction Value for Medical Packaging?
Posted on Leave a Comment
A new study explores the minimum log-reduction value (LRV) for preventing microbial ingress for medical device sterile barrier systems.
Read the full article at: www.packagingdigest.com
Continue Reading »Warning Letter Serves as Reminder to Medical Device Industry
Posted on Leave a Comment

A London, England-based company received an FDA warning letter that should serve as an important reminder for all medical device manufacturers.
Read the full article at: www.mddionline.com
Continue Reading »SeaStar Medical Announces Activation of First Clinical Site in Pivotal Trial with Selective Cytopheretic Device in Critically Ill Adults with Acute Kidney Injury
Posted on Leave a Comment

SeaStar Medical Announces Activation of First Clinical Site in Pivotal Trial with Selective Cytopheretic Device in Critically Ill Adults with Acute Kidney Injury – read this article along with other careers information, tips and advice on BioSpace…
Read the full article at: www.biospace.com
Continue Reading »Peptide-Based Inks for 3D Printing May Boost Regenerative Medicine
Posted on Leave a Comment

Hydrogel could be used for 3D printing structures that can grow active cells for transplant within living beings.
Read the full article at: www.designnews.com
Continue Reading »